Penicillic acid
Penicillic acid
CAS: 90-65-3
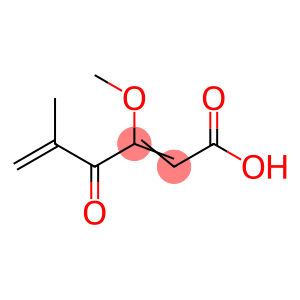
Molecular Formula: C8H10O4
Penicillic acid - Names and Identifiers
Penicillic acid - Physico-chemical Properties
| Molecular Formula | C8H10O4 |
| Molar Mass | 170.16 |
| Density | 1.1456 (rough estimate) |
| Melting Point | 83-87 °C |
| Boling Point | 219.79°C (rough estimate) |
| Solubility | Easily soluble in hot water, ethanol, ether, benzene, chloroform, slightly soluble in cold water (2g/100ml), slightly soluble in hot petroleum ether, almost insoluble in pentane and hexane |
| Appearance | Pale yellow powder |
| Color | Needles from pet ether or rhombic orhexagonal plates |
| Merck | 13,7163 |
| pKa | 3.90±0.33(Predicted) |
| Storage Condition | Store at 2-8 |
| Refractive Index | 1.4425 (estimate) |
| MDL | MFCD00004365 |
Penicillic acid - Risk and Safety
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R22 - Harmful if swallowed |
| Safety Description | S37/39 - Wear suitable gloves and eye/face protection S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S24/25 - Avoid contact with skin and eyes. |
| UN IDs | 2811 |
| RTECS | MM2625000 |
| HS Code | 29189900 |
| Hazard Class | 6.1(b) |
| Packing Group | III |
| Toxicity | LD50 i.p. in mice: 90.00 mg/kg (Chan et al.) |
Penicillic acid - Introduction
Penicillic acid: a Mycotoxin that induces DNA single-strand breaks
Last Update:2022-10-16 17:31:06
Penicillic acid - Reference Information
| (IARC) carcinogen classification | 3 (Vol. 10, Sup 7) 1987 |
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| What is penicillic acid? | penicillic acid, a Penicillium toxin synthesized mainly in Penicillium Mucor, it is mainly produced by the metabolic products of the arc, and the highest content in the feed can reach 2%. Penicillic acid is one of the common mycotoxins that endanger the aquaculture industry. Long-term feeding of feed containing penicillic acid can cause enlargement of animal liver, degeneration of liver cells and inhibit DNA synthesis of animal cells, serious can make the animal cell DNA breakage. Penicillic acid can produce joint toxicity with other mycotoxins, if combined with other mycotoxins can produce greater toxicity. penicillic acid (PA) is a Penicillium toxin found and named by Alsberg and Black(1913), which is mainly synthesized in Penicillium rubra. PA can be detected in many agricultural products, such as corn, dry beans, animal feed and so on. Studies have found that excessive intake of PA can cause a variety of toxic symptoms, including liver toxicity, cytotoxicity and alveolar toxicity, and has carcinogenic effects. The high content of PA in feed, through the enrichment of the food chain, poses a threat to human health. Therefore, it is very important to establish a set of effective rapid detection methods. |
| uses | penicillic acid is mainly a secondary metabolite of Aspergillus and Penicillium, and has a wide range of biological activity. |
| biological activity | Penicillic acid is a polyketide mycotoxin produced by several species of Aspergillus and Penicillium, it is cytotoxic to rat alveolar macrophages (AM) in vitro. Penic acid inhibits Fas ligand-induced apoptosis by blocking caspase-8 self-processing. |
| category | toxic substances |
| toxicity grade | high toxicity |
| Acute toxicity | intraperitoneal-rat LD50 90 mg/kg; Oral-mouse LD50: 600 mg/kg |
| flammability hazard characteristics | flammable, spicy and irritating smoke |
| storage and transportation characteristics | warehouse low temperature, ventilation, dry |
| extinguishing agent | water, carbon dioxide, dry powder, sand |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 20:52:54
Supplier List
Spot supply
Product Name: Benzylpenicillin Impurity 26 Visit Supplier Webpage Request for quotationCAS: 90-65-3
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Penicillic acid Visit Supplier Webpage Request for quotationCAS: 90-65-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: PENICILLIC ACID Request for quotation
CAS: 90-65-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 90-65-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: Penicillic acid Request for quotation
CAS: 90-65-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 90-65-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Penicillic acid Visit Supplier Webpage Request for quotation
CAS: 90-65-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 90-65-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Benzylpenicillin Impurity 26 Visit Supplier Webpage Request for quotationCAS: 90-65-3
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Penicillic acid Visit Supplier Webpage Request for quotationCAS: 90-65-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: PENICILLIC ACID Request for quotation
CAS: 90-65-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 90-65-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: Penicillic acid Request for quotation
CAS: 90-65-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 90-65-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Penicillic acid Visit Supplier Webpage Request for quotation
CAS: 90-65-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 90-65-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History
Penicillic acid
2-methylcyclopropane-1-carbaldehyde
disodium disilicate
Ertugliflozin Intermediate
(1R,2S,5R)-MENTHYL-5R-HYDROXY-[1,3]-OXATHIOLANE-2R-CARBOXYLATE
4-amino-N'-(1-methyl-2-oxo-1,2-dihydro-3H-indol-3-ylidene)benzohydrazide
1795135-61-3
Dimethyl(dimethylamino)(5-cyano-3,3-dimethylpentyl)silane
FORMALDOXIME COLOUR REAGENT
TERT-BUTYL 4-((METHOXYCARBONYL)METHYL)BENZYLCARBAMATE
2-methylcyclopropane-1-carbaldehyde
disodium disilicate
Ertugliflozin Intermediate
(1R,2S,5R)-MENTHYL-5R-HYDROXY-[1,3]-OXATHIOLANE-2R-CARBOXYLATE
4-amino-N'-(1-methyl-2-oxo-1,2-dihydro-3H-indol-3-ylidene)benzohydrazide
1795135-61-3
Dimethyl(dimethylamino)(5-cyano-3,3-dimethylpentyl)silane
FORMALDOXIME COLOUR REAGENT
TERT-BUTYL 4-((METHOXYCARBONYL)METHYL)BENZYLCARBAMATE




